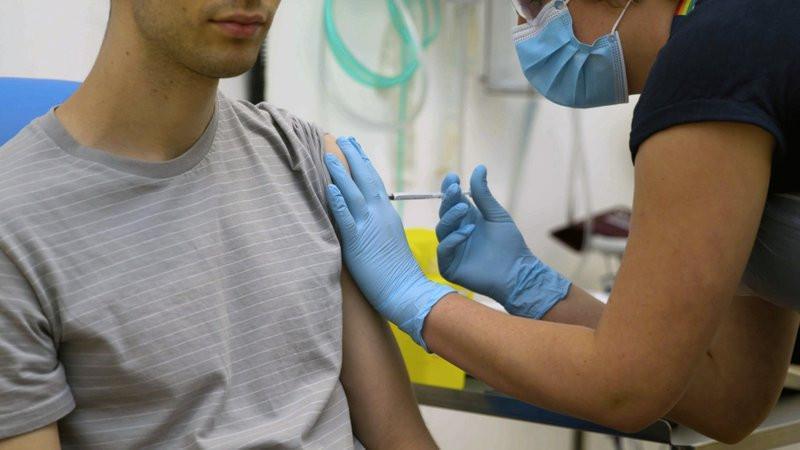
Human trials in the third phase for a possible COVID-19 vaccine being developed jointly by pharma companies BioNTech and Pfizer will take place between September and October in Turkey with 700 volunteers, daily Milliyet has reported.
The companies launched the worldwide phase three for the vaccine in July and Turkey is also taking part in its clinical testing. The potential vaccine will be tested on 30,000 people in 172 countries.
People are already applying to Istanbul University Medical School and Cerrahpaşa Medical School Hospital in Istanbul to volunteer.
The volunteers will be selected from among those who have not contracted the virus and following the antibody tests.
The volunteers should be between the ages of 18 and 65.
“The vaccine will be administered on volunteers within a week. In the next step, data will be collected and analyzed, and the vaccine production may start in November,” Professor Tufan Tükek, the dean of Istanbul Medical School, told daily Milliyet.
Public and private health institutions in Turkey are taking part in the trials, he added.
There will be two groups in the trials, people in the first group will be injected with the vaccine and the second will be the placebo group, according to Professor Dr. İftihar Köksal from the Acibadem Atakent Hospital’s infectious diseases department.